With the comprehensive care programme, we support the Ministry of Health (MoH) in its rollout of protocols reflecting global best practices, as well as introduce healthcare innovations, and promote shorter course TB treatment regimens using new and repurposed drugs.
We have also implemented the TB PRACTECAL clinical trial. This is the first-ever multi-country, randomised, controlled clinical trial, aiming to demonstrate the safety and efficacy of a six-month, all oral treatment regimen for drug-resistant TB. We are now working with the World Health Organization to promote the new regimen as the international standard of care for drug-resistant TB patients.
In the capital, Tashkent, we provide support for the diagnosis and treatment of people living with HIV and co-infections. In 2021, we also launched a mobile laboratory to support the diagnosis of HIV and co-infections among vulnerable people.
Our teams also support the MoH in its efforts to develop the public healthcare system.
Our activities in 2022 in Uzbekistan
Data and information from the International Activity Report 2022.
274
274
€7.8 M
7.8M
1997
1997
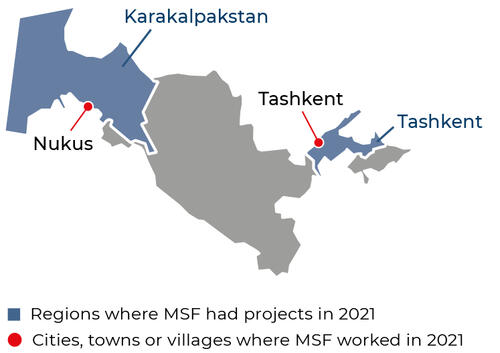

1,550
1,55
Impact of pyrazinamide resistance on multidrug-resistant tuberculosis in Karakalpakstan, Uzbekistan
Pioneering clinical trial for drug-resistant tuberculosis starts

“Please, never give up. You too can beat TB.”
Risk Factors Associated with Default from Multi- and Extensively Drug-Resistant Tuberculosis Treatment, Uzbekistan: A Retrospective Cohort Analysis

Comprehensive TB care for all: The Karakalpakstan Experience